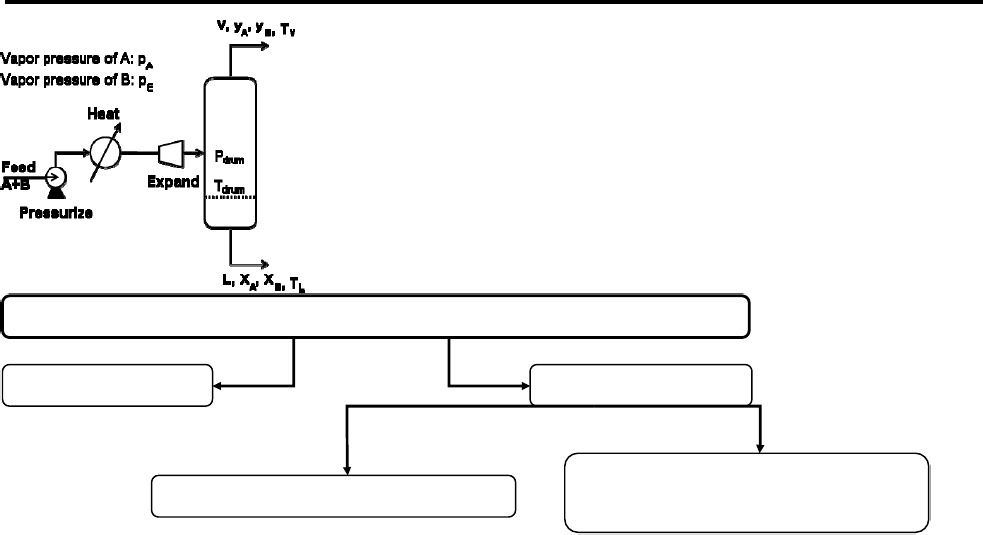
In the enriching section HG = 0.4054 m and HL = 0.253 m, and in the stripping section HG = 0.2835 m and HL = 0.1067 mĪ distillation column with a total condenser and a partial reboiler is separating an ethanol-water mixture. Use a vapor flow rate that is nominally 75% of flooding. We wish to repeat Example 4-3 (distillation of ethanol and water) except that a column packed withĢ-inch metal Pall rings will be used. Equilibrium data for acetone and ethanol at 1 atm Stages, and required q value of the feed. If the feed composition is z = 0.3 (all concentrations are moleįraction of more volatile component), find the optimum feed plate location, total number of The distillation column in parts a and b is separating acetone from ethanol and has xD = 0.9, xB If the bottoms composition is xB = 0.13 mole fraction acetone and theīoilup ratio /B = 1.0, find the vapor composition leaving the second stage above the partialĬ. A distillation column separating acetone and ethanol has a partial reboiler that acts as anĮquilibrium contact. If L/V is equal to 0.8, find the composition of the liquid leaving the fifth stage belowī. A distillateĬoncentration of xD = 0.90 mole fraction acetone is desired. A distillation column with a total condenser is separating acetone from ethanol. If F = 50 mol/h, z = 0.8, p = 1 atm, and ym = 0.892 mole fraction methanol, find V, L, and xm.Ī. If z = 0.4, p = 1 atm, and Tdrum = 77☌, find V/F, xm, and ym.

What must the flow rate and composition of the feed be?į.

We are operating the flash drum so that the liquid mole fraction is 45 mol% methanol. V/F must be used? For a feed rate of 1000 lbmol/h, find product flow rates and compositions.ĭ. If the feed is 30 mol% methanol and we desire a liquid product that is 20 mol% methanol, what Mole fractions and flow rates? Feed rate is 100 kmol/h.Ĭ. Feed is 60 mol% methanol, and 40% of the feed is vaporized. We are separating a mixture of methanol and water in a flash drum at 1 atm pressure. HowĬan you get the composition from P and T? Setting the pressure and temperature also specifies the equilibrium composition. This part is sort of backwards (feed composition is not specified). Which variables have been specified, and how doesĭ. Which two variables (besides the feedĬomposition) have been specified? The drum just separates the vapor from the liquid.Ĭ. The other comments/questions are to help you.Ī. Lesson5_Exercise4_Tom Smith).Problem 2D.1 from your text.
Make sure that your name is in the document title before uploading it to the correct assignment (i.e.Find the Exercise 4 assignment in the Lesson 5 Module by either clicking Next until you find it or by clicking Assignments and scrolling down until you find it.
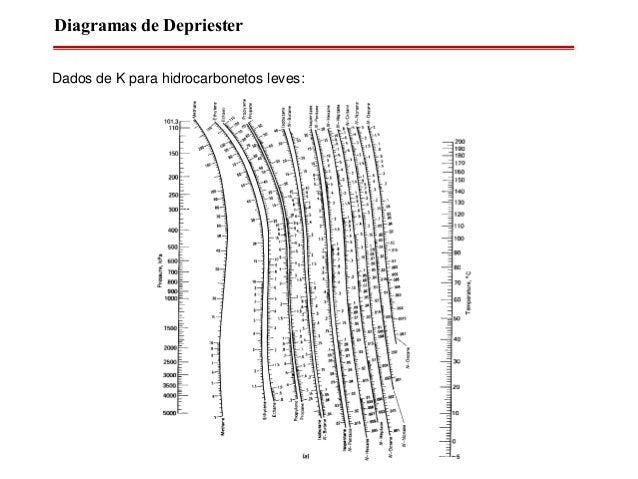
DEPRIESTER CHART K VALUES FOR HYDROCARBONS PDF
Once you have a solution to the exercises you will submit your answers as a PDF by uploading your file to be graded. The rest should be type-written for ease of reading when grading. The scanned pages should relate to using the graphs for solving the problem. Important Note: You may submit scanned images or clear handwritten pages as a PDF that is less than 2 MB in size for this exercise. Submit your answers as a PDF in the Exercise 4 assignment inside the Lesson 5 Module, showing all the steps in your calculations, indicate the K values you read from the nomograms, and state your assumptions, if any. Would the summer, or winter asphalt product be “heavier”? Explain why. Explain which of the following switches will take place in the refinery in about a month, and why? 40 ptsĪ) Switch the deasphalting solvent from propane to pentane.ī) Switch the deasphalting solvent from pentane to propane. In late fall this refinery switches operations to produce more fuel oil from VDR for the coming winter months, producing still some asphalt, but in lower quantity. A refinery in Northwest Pennsylvania produces asphalt as an important product that brings revenue, particularly during the summer months.
